Potential mechanisms underlying ALS are being uncovered at an increasingly rapid rate. Existing medicines targeting them are being repurposed. And, emerging stem cell models of ALS are opening the door to discover new strategies to tackle the disease.
But with the failure of the generic ceftriaxone, Biogen-Idec/Knopp’s dexpramipexole and Trophos’ olesoxime at phase III looming large, key obstacles remain to push potential treatments for ALS forward in the clinic.
At the annual meeting of the American Academy of Neurology (AAN) in Philadelphia this month, experts gathered to discuss emerging strategies currently being evaluated in the clinic. And, key challenges implementing them going forward.
Tirasemtiv
Cytokinetics’ potential muscle booster, tirasemtiv, may help improve breathing and muscle strength of some people with ALS. But tolerability remains a considerable obstacle according to a preliminary analysis of phase IIB clinical trial results presented at AAN 2014.
People taking tirasemtiv appeared to experience one third the reduction of breathing capacity (slow vital capacity) and muscle strength (muscle megascore) compared to those on placebo according to principal investigator Jeremy Shefner MD PhD of the State University of New York.
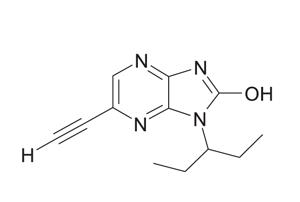
The next Nuedexta? Self-reported outcome measures are now being analyzed to determine whether tirasemtiv may improve the quality of life of some people with ALS.
More than 100 people with ALS, however, could not tolerate tirasemtiv. And, nearly 80 participants of the phase IIB clinical trial dropped out due to gastrointestinal symptoms including nausea and weight loss – three times than those on placebo.
“I think it is clear tirasemtiv has an effect,” says Shefner. “But more work needs to be done to figure out how to deliver the drug in a tolerable way.”
No significant difference in disease progression was detected in people taking tirasemtiv via the ALS functional rating scale, the primary outcome measure of the phase IIB clinical trial. And, no significant improvements in muscle fatigue and other respiratory measures were noted.
Further analysis of the results of the phase IIB clinical trial is ongoing.
“Please understand that this will take time,” says Cytokinetics’ CEO Robert Blum. “We need to delve more deeply into this data before we can commit to what we will be able to do in the next phase.”
To learn more about tirasemtiv including the phase IIB clinical trial, check out our AAN 2014 feature Tirasemtiv: a question of tolerance?
Ozanezumab
GlaxoSmithKline’s ozanezumab may do more than help reconnect motor neurons to muscle fibers in people with ALS according to new results presented by University of California – Los Angeles School of Medicine’s Thomas Carmichael MD PhD at AAN 2014.
The emerging Nogo-A blocker may boost populations of oligodendrocytes – key motor neuron support cells that appear to be lost in people with ALS.
Astrocyte glut? GSK's ozanezumab may increase numbers of oligodendrocytes - helping to refuel motor neurons in people with ALS. Image: Paul Tesar PhD, Case Western Reserve University.
When injured neurons issue an SOS, populations of precursors of oligodendrocytes expand to help repair them.
But according to preclinical studies led by Carmichael, these precursors are unable to transform into these vital support cells –at least after sustaining a stroke.
What’s more, this differentiation block, according to RNA seq analysis, appears to be due to the increased levels of the protein Nogo-A, a key roadblock in the repair of damaged or diseased neurons.
Treatment with the Nogo-A blocker NgR OMNI-Fc increases numbers of mature oligodendrocytes surrounding damaged neurons 4 fold in a mouse model of stroke. And, according to a preliminary analysis, appears to expedite rehabilitation and recovery.
The oligodendrocyte precursors, known as NG2+ cells, are the same cells that accumulate in ALS – at least in a G93A SOD1 mouse model of the disease.
“The Nogo receptor may be sensing the damage – limiting the repair,” says Carmichael.
Oligodendrocytes are emerging to be a key emergency energy source for neurons according to a growing number of studies led by Max Planck Institute of Experimental Medicine’s Klaus-Armin Nave PhD. And, their loss appears to contribute to the onset and progression of the disease according to preclinical studies led by Johns Hopkins University School of Medicine’s Jeffrey Rothstein MD PhD.
A phase II clinical trial of ozanezumab is ongoing. The study is taking place in 11 countries in Asia, Europe and North America. Nearly 300 people with ALS are participating.
The first results could be released as early as the end of 2014.
To learn more about ozanezumab, tune into our podcast with Hôpital de la Pitié-Salpétrière’s Pierre-Francois Pradat MD PhD, A Go for anti-Nogo-A. To find out about other emerging strategies that aim to rebuild muscle- motor nerve connections in people with ALS, check out Micromanaging ALS. To learn more about oligodendrocytes and their emerging role in ALS, tune into our podcast with Klaus Armin-Nave PhD, ALS, Much Ado About Oligodendrocytes.
Neupogen
Serve and neuroprotect? The neuroprotective substance G-CSF may help protect existing motor neurons in people with ALS by reducing inflammation.
Meanwhile, a growing group of neurologists are taking a second look at Amgen’s Neupogen (filagristim) in people with ALS in hopes to slow down their disease.
Neupogen, which contains the neuroprotective substance granulocyte colony stimulating factor (G-CSF), aims to protect motor neurons from further damage by reducing inflammation. The strategy, according to preclinical studies led by Università degli Studi di Milano’s Stefania Corti MD in Italy, may also help create new nerve cells by mobilizing stem cells to the CNS from the bone marrow.
The approach, introduced in the mid 1980s, is currently being used in the clinic to regenerate white blood cells (neutrophils) in bone marrow transplant recipients and cancer patients after chemotherapy.
In 2008, a group of clinicians led by University of British Columbia’s Neil Cashman MD PhD first turned to Neupogen in hopes to treat ALS by replacing neurons destroyed by the disease. The approach aims to deploy stem cells in people with ALS where they are needed – throughout the brain, brainstem and spinal cord.
But although the treatment appeared to mobilize stem cells to the spinal cord (CSF). And, according to results from an open label study of 24 people with ALS led by Università di Torino’s Adriano Chiò MD, reduce the production of key pro-inflammatory substances. The strategy did not improve the survival of people with ALS.
Now, Neupogen is being tested at higher doses administered and/or at higher frequency in hopes to impact progression of the disease.
Increased doses of the drug appear to be safe – at least in the first four people with ALS taking Neupogen participating in an exploratory study led by University of South Florida School of Medicine’s Clifton Gooch MD.
Stemming the tide? ALS clinicians first looked to stem cell therapies to replace motor neurons in people with ALS. But according to some estimates, these strategies could take years to be effective - at least with existing technologies.
Neupogen also appeared to mobilize stem cells - at least into the blood. And, appeared to reduce the production of certain toxic pro-inflammatory substances.
But no effect on disease progression was detected.
A group of clinicians in Germany, however, remains undaunted.
The team, led by Universität Regensburg’s Ulrich Bogdahn MD, Universitätsklinikum Ulm’s Albert Ludolph MD and Medizinische Hochschule Hannover’s Susanne Petri MD, is evaluating the potential treatment strategy in 21 people with ALS taking Neupogen under compassionate use.
People with ALS taking Neupogen appear to show no change in the rate of decline (ALSFRS-R) after one year according to preliminary results from the team presented at AAN 2014 by participating investigator Andrei Khomenko.
But after about 2 years, people taking the drug began to show a small but significant reduction of decline (ALSFRS-R) when compared to historical controls (Pooled Open Access ALS Clinical Trials (ProAct) database). The study remains ongoing.
A clinical trial in Germany is planned for as early as later this year.
Stem cell look book Stem cell therapies aim to protect existing motor neurons in people with ALS by nourishing them and/or detoxifying them. Explore our interactive timeline to learn more about stem cell therapies being developed for the disease.
Neuralstem, Brainstorm and Q-cells
Neuralstem’s potential stem cell strategy NSI-266 for ALS is now at phase II. Brainstorm’s NurOwn is soon to be evaluated at phase II in the US. And astrocyte replacement stem cell-based strategies developed by University of California San Diego School of Medicine’s Lawrence Goldstein PhD / Martin Marsala MD, Johns Hopkins School of Medicine's Nicholas Maragakis MD and Cedar-Sinai’s Clive Svendsen PhD are approaching the IND stage.
But for any of these potential stem cell therapies to truly be useful in the clinic, doctors need to be able track them to make sure these stem cells are settling in (engrafting) in people with ALS where they are needed – throughout the spinal cord.
To meet this challenge, Emory University neurosurgeon Nicholas Boulis MD is turning to ferumoxytol superparamagnetic iron oxide nanoparticles (SPIOs) to tag stem cells before they are transplanted. The strategy hopes Boulis will facilitate the development of potential stem cell strategies for people with ALS – including Neuralstem.
But although this contrast agent appears to detect implanted stem cells. Enable stem cell proliferation and differentiation. And, due to its ultra-small particle size may not affect their migration. Ferumoxytol detects any group of stem cells that engraft into the spinal cord – including those that are dead and therefore of no use to people with ALS.
“SPIOs are more suitable to guide delivery of stem cells to target tissues,” says Johns Hopkins University School of Medicine scientist Amit Srivastava PhD. “And, to monitor engraftment short-term.”
A research team led by Johns Hopkins University School of Medicine’s Jeff Bulte PhD thinks they may have a better solution: 19-fluorine (19F) MRI.
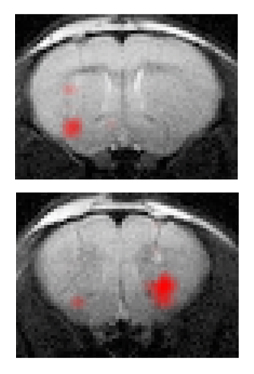
Going on 19? A growing number of researchers are turning to 19F perfluorocarbons to develop potential stem cell therapies - including ALS. The strategy, developed by Celsense Inc, is currently being used in the clinic to develop a potential therapy for certain types of cancers. Image: Neural stem cells (red) transplanted into the mouse brain, Mathias Hoern PhD, PLOS One.
19F MRI tracers can also detect stem cells. And, according to preclinical studies led by Max-Planck-Institut Für Neurologische Forschung’s Mathias Hoehn PhD in Germany and Lunds Universitet’s Zaal Kokaia PhD in Sweden, do not appear to interfere with proliferation, differentiation or migration.
But unlike SPIOs, these contrast agents do not appear to be engulfed by neighboring immune cells according to Srivastava. And, appear to be simply degraded.
The strategy is currently being used to develop potential neural stem cell treatments of a growing number of neurological conditions including stroke.
Now, Srivastava is using these same technologies to develop potential stem cell treatments for ALS in mouse models of the disease.
A key obstacle is that existing MRI tests detecting 19F are slow – meaning more time lying flat – a challenge for many people with ALS. But researchers at Johns Hopkins School of Medicine are working hard to overcome those obstacles.
“Cell tracking with 19F MRI is still in its infancy,” explains Srivastava. “There is a lot of room for improvement.”
To learn more about the ongoing phase II clinical trial of Neuralstem’s potential therapy for ALS, check out Neuralstem phase II begins. To find out more about astrocyte replacement including Q cells, tune into our podcast with Johns Hopkins University School of Medicine’s Nicholas Maragakis MD, A new player in the stem cell Q.