The identification of a common link between repeat expansions in the C9ORF72 gene and ALS one year ago kick-started a race to develop diagnostic tools and therapeutic strategies for this form of ALS. The introduction of antisense treatment strategies however is only one of a number of key advances in ALS that topped the lineup at the 2012 meeting of the Society of Neuroscience (SfN12).
New stem cell technologies open the door toward the identification of new drug targets. New tools enable researchers to discover potential medicines for ALS. And, emerging animal models of ALS might help scientists in future bring more effective treatment strategies to the clinic.
The meeting, which took place in New Orleans, highlighted the latest advances in a wide-range of neurological diseases including ALS. More than 30,000 neuroscientists and neurologists participated.
The livin’ ain’t easy
New models of ALS, unveiled at SfN12, show key signs of ALS opening the door toward the study of sporadic ALS and the development of new strategies to treat the disease. University of California – San Francisco (UCSF) School of Medicine’s Haiyan Qiu PhD introduced a transgenic mouse, harboring an ALS-associated R521C FUS mutation, that showed key ALS symptoms including muscle spasticity and motor deficits. And, the mice sustained a modest but progressive loss of motor neurons in the spinal cord.
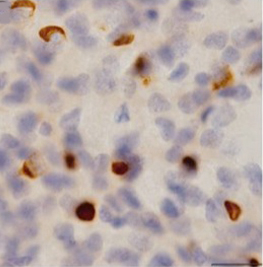
Building up. More than 90% of people with ALS accumulate TDP-43 (brown) in the cytoplasm of neurons in the brain and spinal cord. Models of TDP-43 ALS therefore might provide insight into more cases of the disease. Image: Tsuji, H. et al. Brain (2012). Courtesy of Oxford University Press.
Hoping to bridge the mouse to man gap, Louisiana State University School of Medicine’s Ronald Klein MD PhD is working hard to develop a non-human primate TDP-43 model of ALS. Introduced at SFN12, the rhesus macaques appear to show tell-tale signs of ALS including muscle denervation and deficits completing motor tasks according to a preliminary analysis. “We are trying to make more relevant models,” says Klein, “to discover therapies that we might have [otherwise] missed.”
But key challenges remain. Many models resemble ALS but do not fully replicate the disease. FUS mouse models of ALS developed by University of Massachusetts’ Larry Hayward MD PhD appear to accumulate clumps of FUS protein in the cytoplasm of motor neurons in the spinal cord. But only a subset of these mice exhibit occasional motor coordination deficits and moving difficulties. And, transgenic mutant FUS mice developed by the UCSF team die due to complications outside of the respiratory system including the blood. “We can’t say that they die due to neurodegeneration,” says Qiu.
What’s more, some models of ALS show few signs of the disease. A ubiquillin 2 mouse model of ALS according to results presented by Northwestern University School of Medicine’s Han-Xiang Deng MD PhD accumulates clumps of proteins largely in the brain - exhibiting behavioral challenges resembling frontotemporal dementia (FTD). “We waited two years,” says Deng. “We didn’t see a motor phenotype.”
Laissez les cellules souche rouler
Stem cells may be a promising alternative to study ALS and discover new drugs for the disease according to some experts.
Induced pluripotent stem (iPS) cell-derived motor neurons appear to exhibit tell-tale signs of ALS according to researchers from the lab of Harvard University’s Kevin Eggan PhD. The motor neurons, generated from skin biopsies from 2 people with SOD1-linked ALS, accumulate aggregate-destroying vacuoles and appear to be more vulnerable to degeneration according to Harvard University’s Evangelos Kiskisnis PhD. The mitochondria of these cells also appear to be malfunctioning (swollen) and struggle to get around the axon. Such power plant delivery interruptions, first observed in mouse SOD1 models of ALS, are speculated by scientists to contribute to the unplugging of the motor nerves from skeletal muscle in a growing number of motor neuron diseases.
“Induced pluripotent stem cells are a robust, reliability technology to study ALS,” says Kiskisnis.
What’s more, these cultured motor neurons appear to fire spontaneously and more frequently according to electrophysiological patch clamp analysis presented by Harvard University neuroscientist Brian Wainger MD PhD. Extracellular arrays of these motor neurons enable 100s of these cells to be characterized simulataneously according to Wainger - opening the door to screen for drugs that could reduce this so-called hyperexcitability – one of the earliest signs of the disease.
Creating motor neurons from induced pluripotent stem cells, however, takes time and can be tricky to do – particularly at large-scales needed to discover potential medicines for ALS. So, Harvard University’s Justin Ichida PhD is hoping to use a shortcut – create motor neurons directly from skin biopsies and screen for substances that the protect them from the damage that occurs due to the disease. A pilot screen, presented at SfN12, identified one potential drug, kenpaullone, which appears to boost survival of these so-called ALS induced motor neurons.
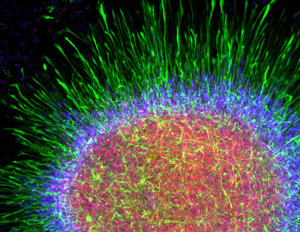
Search and destroy. Scientists hope to identify new ALS targets by uncovering astrocytes' weapons of motor neuron destruction in iPS-based models of ALS. Image: Courtesy of Robert Krencik, University of Wisconsin-Madison.
Meanwhile, other research teams are working to recreate ALS in dishes by introducing other cells such as astrocytes and microglia and studying their weapons of motor neuron destruction. The results might help researchers identify new targets that can be used to identify potential medicines for the disease.
New methods might help scientists regenerate these so-called glial cells from people with ALS to kickstart studies of the disease. A technique developed by the team of Nationwide Children’s Hospital’s Brian Kaspar MD PhD enables ALS-ravaged astrocytes to be made more quickly – by directly generating them from skin biopsies. And, a method developed by the lab of University of Rochester’s Steven Goldman MD PhD generates myelinating-capable oligodendrocytes from human iPS cells helping researchers to unravel their emerging role in the disease.
What’s more, the first fully humanized stem cell-based model of ALS developed by the team of Columbia University’s Serge Przedborski MD PhD opens the door toward unraveling underlying mechanisms of the human disease. “We can really recapitulate in a dish,” says Columbia University neuroscientist Virginia Le Verche PhD, “what we see in patients.”
But the jury is still out whether stem-cell based strategies can be used to treat ALS. The transplantation of neural stem cells into the cervical (diaphragm-moving) region of the spinal cord appears to be safe according to preclinical studies from the research team of Cedars Sinai Medical Center’s Clive Svendsen PhD. But these strategies do not appear to improve muscle function or increase survival – at least in a SOD1–linked rat model of ALS. The findings add to growing evidence which suggests that multiple injections into multiple regions might be needed to maintain the connections between the brain, descending motor tracts and lower spinal cord to keep muscles moving. “All of these structures need to be functional and connected properly," explains University of California San Diego's Martin Marsala MD, “in order for people to walk.”
Tech po’boy
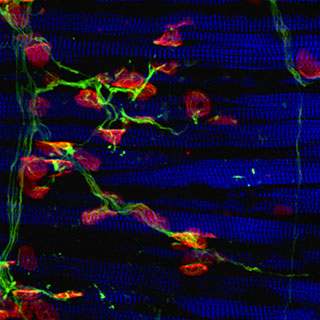
Structural support. A new technique might enable scientists to identify potential ALS medicines that stabilize muscle-motor nerve connections - possibly slowing the onset of muscle weakness and paralysis. Image: Rockefeller University.
Researchers, however, continue to introduce new tools to discover and develop new potential treatment strategies for ALS.
Neuromuscular junctions (NMJs) can now be recreated in the laboratory paving the way toward the discovery of new ALS medicines that protect and stabilize these connections. The technique, developed in the lab of Dalhousie University’s Victor Rafuse PhD, enables stem cell-derived motor neurons to plug directly into muscle fibers – just like in our muscles. What’s more, connections recreated between SOD1 ALS motor neurons and muscle cells appear to be much more unstable. Existing techniques regenerate NMJs using cultured muscle cells. Whether these motor neurons retain their muscle moving abilities (to fire action potentials) remains to be determined.
Protein aggregate clearance via autophagy can now be monitored in single neurons - opening the door toward ALS “clinical trials in dish” according to UCSF’s Steve Finkbeiner MD PhD. Initial studies identified four destruction-stimulating substances which increase the survival of motor neurons recreated from skin cells from people with ALS. These so-called autophagy inducers appear to protect neurons at least in part, by reducing a potential key source of toxicity associated with the disease. “We not only improve survival,” says Finkbeiner, “we seem to be able to accelerate the clearance of TDP-43.”
What’s more, this drug discovery strategy is potentially promising for a growing number of neurodegenerative diseases including ALS. The inability to destroy misfolded proteins through these mechanisms appears to be a key contributor to the disease according to new studies presented by Northwestern University School of Medicine’s Teepu Siddique MD and his team. “If we can modulate proteostasis, we may be able to convert a fatal disease into a long term disability,” says Siddique.
Meanwhile, University of Missouri-Columbia speech pathologist Teresa Lever PhD is developing quantitative tests to help diagnose and treat dysphagia (difficulties swallowing). Existing techniques rely on qualitative approaches or are measured on a self-reporting scale. The tests, which include fluoroscopic, histological and video analysis, are currently being evaluated by the University of Missouri team in animal models of ALS. Looking ahead, these methods might help scientists discover and develop the next Nuedexta, a medicine that is soon to be tested in the clinic for its ability to improve speaking and swallowing in people with ALS.
* * *
To learn more about how scientists hope to use stem cells to discover new therapies for ALS, read: iPS, ready, set screen. To find out more about the challenges of using stem cells to treat ALS, check out Neuralstem, surging immunosuppression? To learn more about treatment strategies currently being developed for C9ORF72-linked ALS, tune into our podcast with Jeff Rothstein MD PhD, ALS dressed to the C9s.