A disruption in the matrix? MMP-9 may contribute to the destruction of motor neurons in part by degrading laminins, ECM components that may be important in stabilizing neuromuscular junctions. Image: Originally published in ASBMB Today. David Goodsell: The master of mol art. Sergei Shukov, August 2011. ©The American Society for Biochemistry and Molecular Biology.
In 1995, the first drug for ALS, riluzole, hit pharmacy shelves. The drug extends survival about 3 – 6 months. But nearly twenty years later, riluzole remains the only drug approved to treat the disease.
The reason, in part, is that researchers remain unsure why motor neurons are selectively destroyed in ALS. And, therefore how to best protect them from the onslaught of the disease.
In hopes to overcome these challenges, Columbia University’s Christopher Henderson PhD turned to gene profiling in G93A SOD1 mice to identify proteins that may make motor neurons vulnerable to ALS by looking for substances strongly reduced or absent in cells unaffected by the disease.
Now, the Columbia University team reports that the enzyme matrix metalloproteinase 9 (MMP-9) may make motor neurons susceptible to ALS. And, trigger their destruction.
Motor neurons producing high levels of MMP-9 appear to be selectively destroyed by ALS. And, the removal of the enzyme delays the onset of paralysis and extends survival 25%.
“It may explain why fast motor neurons are vulnerable,” explains Henderson. “That’s something that is common to all forms of ALS.”
The results suggest that MMP-9 may be a key contributor to ALS. And, reducing the enzyme may be a potential strategy to slow the progression of the disease.
Life of MMPIs
Clinicians first looked to MMPIs in the 1980s in hopes to stop cancer in its tracks. But over the years, matrix metalloproteinases emerged as a key instigator of neuronal destruction. And, these experimental medicines became recruited in an even larger war: stroke, spinal cord injury (SCI), traumatic brain injury (TBI) and neurodegenerative disease.
Matrix metalloproteinases appear to rise up in the central nervous system in a number of neurological conditions leading to blood brain barrier breakdown, infiltration of T cells, monocytes and macrophages, and neuroinflammation.
Some of the same mechanisms that ultimately destroy motor neurons in people with ALS.
An oracle of the disease? The levels of MMP-9 appear to correlate with the progression rate of ALS suggesting that the enzyme may play a role in the disease.
MMP-9 appears to be in the right place at the right time to contribute to ALS. The enzyme builds up in the motor centers of the brain and spinal cord according to studies led by University of Southern California's Zoltan Tökés PhD And, the levels of MMP-9 appear to correlate with the rate of progression of the disease according to studies led by Universitätsklinikum Ulm's Johannes Brettschneider MD.
What’s more, according to preclinical results from University of Arkansas College of Medicine’s Mahmoud Kiaei PhD, these enzymatic uprisings appear to break out in motor neurons –key cells ravaged by ALS. And, in microglia, key instigators of inflammation, which fuel the progression of the disease.
”MMP-9 could be an important player that could contribute to motor neuron death,” says Kiaei. "It is an exciting molecule that could allow us to learn a lot about ALS."
Now, MMP-9 is fingered as a key player in the destruction of key subsets of motor neurons in ALS – at least in mice.
Motor neurons producing high levels of MMP-9 appear to be preferentially lost during the course of the disease. And, increasing the levels of the enzyme in motor neurons appeared to accelerate their destruction.
A radical architect at work? Free radicals and inflammatory substances released by microglia may activate MMP-9 causing activated enzyme levels to build up during the course of the disease. Image: Urs Meyer MD, Swiss Federal Institute of Technology.
What's more, re-introducing MMP-9 into G93A SOD1 mice lacking the enzyme appeared to increase their vulnerability to the disease.
“Other motor neurons will eventually be destroyed in ALS,” explains Henderson. “We are talking about the first motor neurons to go.”
The results suggest that MMP-9 may mark motor neurons for destruction in people with ALS. And, reducing levels of the enzyme may protect them from the disease.
ALS Potion Number 9?
But targeting MMP-9 is tricky to do.
Research teams developed more than 50 matrix metalloproteinase inhibitors to date. All of these experimental medicines appear to be intolerable. And, all of them appear to be ineffective in treating cancer, rheumatoid arthritis and heart disease.
“Matrix metalloproteinases are a bit like zombies. They resurface again and again from pharmaceutical graveyards.” says ALS Therapy Development Institute’s Matvey Lukashev PhD. “But without much success.”
The reason, in part, is that most of these experimental medicines target many matrix metalloproteinases.
Enzymes that are essential to build new blood vessels and repair muscles – critical in people with neurological conditions including ALS.
"Unfortunately, the inhibitors that we need are not out there yet," says University of Arkansas College of Medicine's Mahmoud Kiaei PhD.
Dodging bullets? Emerging inhibitors of gelatinases may help protect motor neurons in ALS by reducing levels of MMP-9 - an enzyme that may make them vulnerable to the disease. Image: Clive Svendsen PhD, Cedars-Sinal Medical Center. ©University of Wisconsin-Madison University Communications.
However, inactivating MMP-9 alone in people with ALS is extremely difficult to do.
More than 20 matrix metalloproteinases have been discovered to date. All of these enzymes are highly structurally related. And, many of them are produced in the brain and spinal cord.
What’s more, many of these matrix metalloproteinases use the same strategies to remodel and repair our tissues.
But University of Notre Dame chemists Shahriar Mobashery PhD and Mayland Chang PhD remain undaunted.
In 2000, Mobashery’s team introduced SB-3CT, a drug that targets gelatinases: matrix metalloproteinase 2 and 9. The drug, known as a suicide substrate, binds deep in the active site of these enzymes – keeping them out of action.
SB-3CT, redesigned and reimagined by Chang’s team, can now be directly introduced and absorbed into the blood. And, appears to be readily delivered into the central nervous system.
The strategy is now being developed as a potential treatment for traumatic brain injury and stroke.
“We are trying to get this drug into the clinic,” says University of Notre Dame’s Mayland Chang PhD.“We think it could be beneficial for a number of neurological diseases.”
The drug, according to preclinical studies led by University of Missouri School of Medicine’s Zezong Gu MD PhD, appears to protect damaged neurons from degenerating. And, stabilizes the vasculature after traumatic brain injury and stroke.
The potential treatment strategy, however, may do much more for people with ALS.
The emerging gelatinase blocker appears to decrease infiltration of activated monocytes, macrophages and T cells – emerging instigators of inflammation, the fuel that drives the progression of the disease.
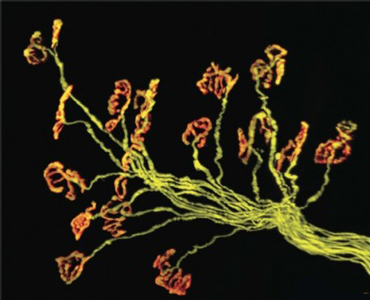
A matrix reboot? Emerging gelatinase inhibitors may need to be delivered acutely to enable the repair of injured motor neurons and re-innervation into muscle. Image: Greg Valdez PhD, now at Virginia Tech Carilion Research Institute and Joshua Sanes PhD, Harvard Medical School.
Treatment with SB-3CT appears to lower numbers of monocytes and macrophages that invade the injured spinal cord about 30% according to preclinical studies led by University of San Francisco’s Linda Noble-Hausslein PhD. And, significantly reduce effector T cell infilitration– at least in a model of transplant rejection according to studies led by Indiana University David Wilkes MD.
What’s more, the emerging gelatinase blocker may supe up regulatory T cells, key immune cells that help keep ALS in check early in the disease. And, boost numbers of oligodendrocytes – an emerging life-line for motor neurons disrupted in the disease.
But how to use these emerging medicines to regulate levels of MMP-9 in people with ALS remains an open question.
MMP-9 appears to contribute to the destruction of motor neurons. But these same enzymes may ultimately be needed to enable recovery from the disease.
Matrix metalloproteinases appear to facilitate the repair and regeneration of axons – a key first step to plug motor neurons back into muscles.
Enzymes including MMP-9 appear to degrade key sugar-coated proteins in the surrounding debris known as chondroitin sulfated proteoglycans (CSPGs) that block the regrowth of axons. And, according to preclinical studies, at least MMP-2 is required to repair damaged motor neurons in the spinal cord.
Emerging gelatinase inhibitors may therefore need to be administered acutely at the right place and at the right time during the course of the disease.
A similar strategy is being developed to treat traumatic brain injury, spinal cord injury and stroke.
"There is no such thing as a safe drug," says Chang. "There is always a balance between risk and benefit."
Morphing MMPIs
Matrix metalloproteases are beginning to emerge as a key player in a growing number of neurological conditions – including ALS.
Motor neurons that produce these enzymes appear to be selectively destroyed by ALS. And, microglia fuel the MMP9 outbreak – leading to blood brain barrier breakdown, infiltration of immune cells, inflammation and motor neuron loss.
The blue or the red pill? Reduction of MMP-9 may be one of many strategies needed to treat ALS. Image: Fernando Prieto, Flickr.
Reducing MMP9 may be a potential strategy to slow progression of the disease. But how to quell these enzymatic uprisings in people with ALS remains an open question. Most MMP-targeted medicines tested in the clinic to date appear to be intolerable. And, painful side effects preclude their long-term use.
Pharmaceutical companies, burned by multiple failures, appear to be closing the MMP books. But researchers remain undaunted. Emerging sub-selective inhibitors of gelatinases appear to protect neurons and reduce inflammation – at least in mice. And, strengthen the vasculature.
Their short-term use, however, in combination with other therapies, may be necessary to enable the repair of injured motor neurons and reconnect them to muscles.
By understanding how MMP-9 is activated and the role of the enzyme in the disease, researchers hope to develop more effective treatments for ALS. And, encourage industry leaders to repurpose existing experimental medicines already in their freezers.
“I think MMP-9 is one of a small number of targets," says Henderson, "that is definitely worth further investigation in clinical trials."
References
Kaplan, A., Spiller, K.J., Towne, C., Kanning, K.C., Choe, G.T., Geber, A., Akay, T., Aebischer, P. and Henderson, C.E.. (2014) Neuronal matrix metalloproteinase-9 is a determinant of selective neurodegeneration. Neuron 81(2), 333-348. Abstract | Full Text (Subscription Required)
Lim, G.P., Backstrom, J.R., Cullen, M.J., Miller, C.A., Atkinson, R.D. and Tökés, ZA. Matrix metalloproteinases in the neocortex and spinal cord of amyotrophic lateral sclerosis patients. Journal of Neurochemistry 67(1), 251-259. Abstract | Full Text (Subscription Required)
Fang, L. et al. (2009) Linking neuron and skin: matrix metalloproteinases in amyotrophic lateral sclerosis (ALS). Journal of Neurological Sciences 285 (1-2), 62-66. Abstract | Full Text (Subscription Required)
Kiaei, M., Kipiani, K., Calingasan, N.Y., Wille, E., Chen, J., Heissig, B., Rafii, S., Lorenzl, S. and Beal, M.F. (2007) Matrix metalloproteinase-9 regulates TNF-alpha and FasL expression in neuronal, glial cells and its absence extends life in a transgenic mouse model of amyotrophic lateral sclerosis. Experimental Neurology 205(1), 74-81. Abstract | Full Text (Subscription Required)
Lorenzl, S., Narr, S., Angele, B., Krell, H.W., Gregorio, J,. Kiaei, M., Pfister, H.W. and Beal, M.F. (2006) The matrix metalloproteinases inhibitor Ro 28-2653 extends survival in transgenic ALS mice. Experimental Neurology 200(1), 166-171. Abstract | Full Text (Subscription Required)
Song, W. et al. (2013) Water-soluble mmp-9 inhibitor prodrug generates active metabolites that cross the blood-brain barrier. ACS Chemical Neuroscience 4(8), 1168-1173. Abstract | Full Text (Subscription Required)
Brown, S., Bernardo, M.M., Li, Z.-H., Kotra, L.P., Tanaka, Y., Fridman, R. and Mobashery, S. (2000) Potent and Selective Mechanism-Based Inhibition of Gelatinases. Journal of the American Chemical Society 122:6799–6800. Abstract | Full Text (Subscription Required)
Further Reading
Vandooren, J., Van den Steen, P.E. and Opdenakker G. (2013) Biochemistry and molecular biology of gelatinase B or matrix metalloproteinase-9 (MMP-9): the next decade. Critical Reviews of Biochemistry and Molecular Biology 48(3), 222-272. Abstract | Full Text (Subscription Required)
Zhang, H., Chang, M., Hansen, C.N., Basso, D.M. and Noble-Haeusslein, L.J. (2011) Role of matrix metalloproteinases and therapeutic benefits of their inhibition in spinal cord injury. Neurotherapeutics 8(2), 206-20. Abstract | Full Text