
Irreconcilable differences Key genetic changes called modifiers may explain why some people with ALS may get the disease later or live longer. Image: Kam2y, Flickr.
Many people with ALS survive 3-5 years. But at least 10% of people with ALS live for more than 10 years. And, many of them harbor the same mutations in the same genes.
"There is no straight line between cause and clinical outcome in ALS," says University of Leuven's Wim Robberecht MD PhD.
The reason, in part, is that key changes buried in their genomes may determine when the first signs of ALS appear and the progression rate of their disease.
Identifying these genetic modifiers may enable researchers to pinpoint key targets of ALS. And, develop more effective treatments for the disease.
Ephrin receptor A4 (EphA4), an emerging modifier of ALS, appears to be upregulated in people with rapidly progressing ALS. And, lowering its production, at least in two people with ALS, may explain in part, their extended survival after being diagnosed with the disease.
The discovery, reported by Wim Robberecht MD PhD in 2012, suggested that reducing ephrin signaling may be a potential treatment strategy for ALS.
This month, researchers gathered at the 2013 meeting of the Society of Neuroscience (SfN13) in San Diego to discuss the emerging role of ephrins in neurological conditions including ALS. And, the latest ephrin blockers being developed to treat them.
Gone Fishing
Researchers across the globe are working hard to decode the genomes of 1000 people with ALS. The multi-institutional partnership, led by HudsonAlpha’s Rick Myers PhD, in part, aims to ferret out modifiers of ALS in hopes to identify new treatment strategies for the disease. The project is expected to take about 5 years.

Go fish Scientists cast out for modifiers of ALS by looking for genetic changes in a zebrafish that protected them from developing key features of the disease. Image: Carnegie Mellon University.
Wim Robberecht MD PhD, however, chose a simpler approach. He sent his research team fishing.
The University of Leuven team developed a zebrafish SOD1 model of ALS. And, screened for key genetic changes that protected the fish from developing key aspects of the disease.
Their biggest catch: EphA4.
The reduction of EphA4 appeared to slow ALS – at least in mice. A mouse model of ALS that produced 50% of normal levels of EphA4 appeared to experience about a 40% reduction in decline in motor coordination/performance. And, the survival of these mice appeared to be extended more than 50% after showing the first signs of disease.
What’s more, reduced expression of EphA4 may be a key compensatory mechanism that may extend the survival of some people with the disease. People that expressed lower levels of EphA4 appeared to live longer with ALS. And, two of them harbored key mutations in the EphA4 receptor gene.
But why levels of EphA4-mediated signaling impacted the outcome of people with ALS remained unclear.
Now, neuroscientist Lies Schoonaert of the University of Leuven reports that ephrin B2, a protein which binds and activates EphA4, may in part, explain its role in modifying the disease.
Ephrin B2 appears 'to relocate' to astrocytes after disease onset in a G93A SOD1 mouse model of ALS. And, fuel the progression of the disease.
G93A SOD1 mice harboring activated astrocytes lacking ephrin B2 appear to progress more slowly. And, live more than two weeks longer with the disease.
The results follow at the heels of a 2013 study led by University of Rochester School of Medicine’s Maiken Nedegaard MD DMSc which fingered ephrin B2 as a key obstacle blocking the repair and regeneration of axons in a mouse model of spinal cord injury.
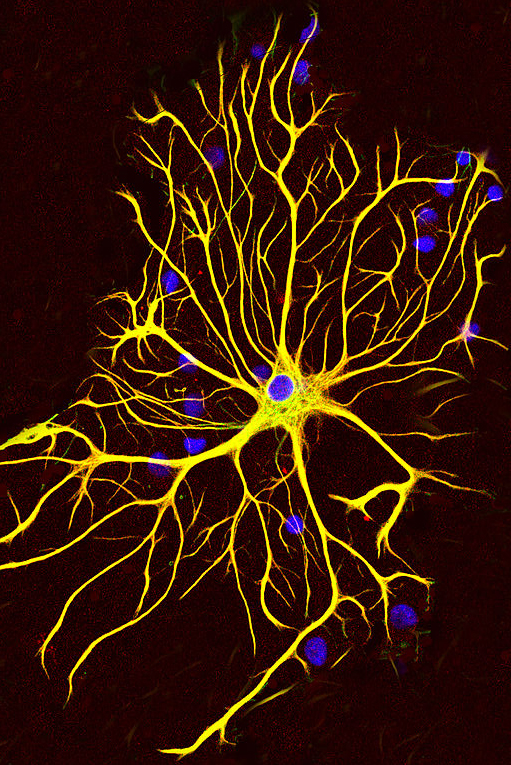
Signals crossed? Researchers suspect that ephrin B2 produced by reactive astrocytes (above) binds ephA4 decorating motor neurons - contributing to ALS. Image: Gerry Shaw, Wikimedia Commons.
Together, the results suggest that ephrin-B2 may prevent the repair of motor neurons damaged by ALS– accelerating the progression of disease.
Now, Robberecht’s team hopes to identify key downstream targets of ephrin B2 in motor neurons in hopes to identify new targets of ALS. And, develop more effective treatment strategies for the disease.
Check Angiogenesis’ List
Sanford Burnham Medical Research Institute’s Elena Pasquale PhD suspected long ago that targeting EphA4 may help stop cancer in its tracks.
The activation of EphA4 triggers the growth of new blood vessels – fueling the growth of tumors. And, accelerates its spread.
In more recent years, however, this strategy is emerging as a potential treatment for a wide range of neurological conditions – including ALS.
The reason, according to a growing number of studies, is that EphA4-mediated signaling appears to be a key barrier blocking the repair of damaged nerve cells in the brain and spinal cord.
EphA4 blockers and decoys may help lift this ‘brake’ in people who suffered a spinal cord injury or stroke - enabling the wiring to be repaired and reconnected. And, in people with ALS, these ‘antagonists’ may help the motor nerves plug back into muscles.
But targeting EphA4 is tricky to do.
EphA4 plays a number of key roles – including helping to keep bones healthy, regulate glucose intake and maintain the blood supply to the brain, heart and lungs.
“Targeting ephrin is hard,” says ALS Therapy Development Institute’s Steve Perrin PhD. “There is a lot to think about when converting it into a clinical candidate for ALS.”
EphA4 blockers may need to be targeted to key tissues affected by ALS to minimize side effects.
What's more, potent highly-selective small molecule blockers continue to remain elusive according to Sanford-Burnham Medical Institute’s Elena Pasquale PhD. And, existing peptides that target EphA4 are extremely instable- insufficient for therapeutic use.
To overcome these obstacles, Elena Pasquale PhD turned to Sanford-Burnham structural biologist Stefan Riedel PhD in hopes to design more stable EphA4 blockers that could potentially be used in the clinic.
The plan: Take snapshots of peptide-bound EphA4 at molecular resolution. By understanding how these peptides grab hold of EphA4, the team could get a better sense of their plan of attack. And, design and deploy more effective ones that keep EphA4 out of action.
The Sanford-Burnham team captured EphA4 bound to three peptides, APY, KYL and VTM at atomic resolution. By taking a look at these molecular snapshots, the team found that these peptides burrowed into EphA4. And, blocked its ability to bind ephrins - including potentially ephrin-B2.
But Scripps Research Institute synthetic chemist Philip Dawson PhD saw room for improvement. The peptides, he reasoned based on computational calculations, could be redesigned and reimagined to be both stable and more effective.
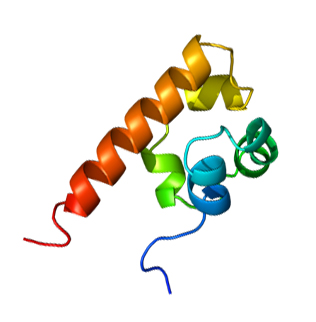
Be rational Researchers took a snapshot of EphA4 bound to APY in hopes to design cyclic peptides more suitable for therapeutic use.
Under the laboratory hood, Dawson’s team created cyclic peptides that bound more tightly to EphA4. And, subsequently modified them to boost their stability.
One highly selective cyclic peptide, called APY-d3, appears to be potent (IC50 = 27 ± 6.5 nM). And, “extremely stable” both in CSF and in circulation (t1/2 > 72 hours) - at least in mice.
The cyclic peptide was introduced just one day after State University of New York's Jing An MD PhD unveiled TYY, an EphA4-targeted cyclic peptide being developed by her team as a potential treatment for certain cancers.
Now, Pasquale’s team hopes to develop this cyclic peptide-based strategy as a potential treatment for neurological conditions including ALS.
Key obstacles remain. These peptides needs to be chemically modified to enable delivery into the brain. And, maximize its stability in circulation.
But Elena Pasquale PhD remains hopeful that her team can overcome these challenges.
“Cyclic peptides are a particularly promising approach,” says Pasquale.
Signal Ahead
The Ephrin A4 receptor (EphA4) is emerging as a key regulator of the regeneration of the central nervous system. And, a formidable obstacle in the repair of the brain and spinal cord upon injury or disease.
Increased Eph/ephrin signaling is implicated in a growing number of neurological conditions including ALS. And, may accelerate the progression of the disease.
What’s more, reduced EphA4 signaling may be a potential compensatory mechanism that may help protect the motor nerves in some people with the ALS. Reducing this signaling with emerging EphA4 antagonists and decoys may be helpful to extend survival of people with the disease.
But key challenges remain. EphA4 blockers may need to be targeted to key systems affected by ALS to minimize side effects. And, modified to ensure delivery to many of these tissues – including the brain and spinal cord.
Key downsteam targets of Eph/ephrin signaling also need to be elucidated according University of Melbourne’s Ann Turnley PhD. Identifying these targets will enable scientists to better understand why the nervous system ‘hits’ the brakes when damaged. And, create treatment strategies to lift them to allow repairs to take place.
“There’s a lot of work that still needs to be done. But the future looks promising,” says Ann Turnley PhD.
References
Van Hoecke, A. et al. (2012) EPHA4 is a disease modifier of amyotrophic lateral sclerosis in animal models and in humans. Nature Medicine 18(9), 1418-1422. Abstract | Full Text (Subscription Required)
Ren, Z., Chen, X., Yang, J., Kress, B.T., Tong, J., Liu, H., Takano, T., Zhao, Y. and Nedergaard, M. (2013) Improved axonal regeneration after spinal cord injury in mice with conditional deletion of ephrin B2 under the GFAP promoter. Neuroscience 241, 89-99. Abstract | Full Text (Subscription Required)
Han, X., Xu, Y., Yang, Y., Xi, J., Tian, W., Duggineni, S., Huang, Z. and An, J. (2013) Discovery and Characterization of a Novel Cyclic Peptide That Effectively Inhibits Ephrin Binding to the EphA4 Receptor and Displays Anti-Angiogenesis Activity. PLoS One 8(11), e80183. Abstract | Full Text
Further Reading
Noberini, R., Lamberto, I. and Pasquale, EB. (2012) Targeting Eph receptors with peptides and small molecules: progress and challenges. Abstract | Full Text