Recently, researchers at the Oregon State University (OSU) published results from their preclinical experiments of a molecule called copper-ATSM (CuATSM) in an ALS mouse model. Those results were positive, and the researchers’ data suggests that treatment with this molecule in mice halted the disease’s progression. This is a summary of those results and information known about the potential of CuATSM as a potential treatment for ALS.
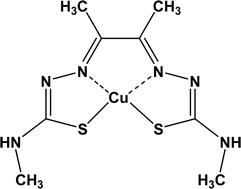 What is CuATSM?
What is CuATSM?
CuATSM (diacetyl-bis(4- methylthiosemicarbazonato)copperII) (pictured) is a molecule which for many years has been used in imgaging experiments to locate tissues within a person’s body that aren’t receiving sufficient oxygen (hypoxic tissues). With a lack of oxygen, scientists have also found that these tissues have damaged or dysfunctional mitochondria. While CuATSM has been mostly used in imaging technologies up to this point, neuroscientists became interested in CuATSM as a potential therapeutic agent after they found it has the ability to deliver much needed copper safely to specific cells within the body which may use that copper to prevent the malformation of proteins known to cause neurodegenerative disorders, such as ALS.
In some ALS cases, one of the things found to be going wrong is that cells are producing a misfolded version of the SOD1 protein, often due to a genetic mutation in the SOD1 gene itself. In addition, research from many labs around the world have also found mutated SOD1 protein in sporadic cases of ALS and suggest that perhaps the mutant protein spreads (in a similar way to prion diseases) from cell to cell. CuATSM has been shown in laboratory-based studies to deliver copper to cells which are producing this misfolded protein. Specifically, a molecule within human cells called copper chaperone for SOD (CCS) will transfer the copper from CuATSM and use it to help prevent the misfolding of the SOD1 protein.
What was reported in the paper?
This is not the first study of CuATSM in ALS mice. Previous work has been published by the authors at OSU as well as from investigators at the Florey Institute of Neuroscience and Mental Health at the University of Melbourne in Australia. However, this paper does produce unique results which are important to the understanding and potential advancement of CuATSM as a therapeutic opportunity for ALS and other neurological disorders. Primarily, there are two important findings: that a mouse model was created to express effectively mutant SOD1 protein and CCS which mimics some aspects of human ALS of ALS, and secondly, that treatment of CuATSM within the model halts the disease progression.
CCS is a necessary and abundant molecule found in humans, however it is not found in mice. The mutant SOD1 protein associated with some forms of ALS similarly is a human phenomenon. As such, the levels of mutant SOD1 and CCS needed to conduct the appropriate preclinical experience are not natively found in mice, therefore researchers genetically engineered a mouse model that does. Untreated, the SODG93AxCCS animals died within the first several weeks of life.
A total of 45 SODG93AxCCS mice were reported on by the OSU scientists in the published paper. That included four separate cohorts of mice in the experiment ranging from no treatment with CuATSM, intermittent treatment and continuous CuATSM treatment. Mice treatment intermittently showed a slowing of disease progression during treatment and a return to decline when treatment was removed. Continous treatment from day 5 of life on in the SODG93AxCCS model produced the most favorable results, with those animals living an average of 600 days of about 30 times longer than those animals that received no treatment at all. Researchers at OSU also conducted an experiment using regular SODG93A mice. In that experiment they saw an extension of life as well, both when starting dosing with CuATSM when the mice were either 5 or 50 days old, although a much higher dose of CuATSM was used in these experiments than in the cross model.
Scientists from OSU show, as others have before them, that CuATSM gets into the spinal cord of mice, where it is thought most needed to address the issue posed by the misfolding of SOD1 protein which has been implemented in the development of some forms of both genetic and sporadic ALS.
Bottom Line
The ALS Therapy Development Institute has been aware and following the CuATSM story in ALS for a number of years. We are encouraged by the results from Oregon State, which builds on work published by others investigating the potential application of CuATSM in ALS. However, as the authors of this paper have noted in the media, people with ALS shouldn’t just start ingesting a bunch more copper, as that approach isn’t the same as the one being approached using CuATSM.
The path toward the clinic for CuATSM may be challenging, as it is currently mostly used as a marker for imaging, not as a therapeutic, so dosing of the compound in people for therapeutic potential still requires additional investigation. However, as CuATSM has a long history of safe use in the imagining of hypoxic tissue in people, there is an opportunity for rapid clinical development. More work is needed in the lab before those trials would start. Researchers at OSU and elsewhere have stated in the media that they are working toward that end now.
A company called Procypra Therapeutics (aka Collaborative Medicinal Therapeutics) has plans to file the paperwork required to initiate a clinical trial of their derivative form of CuATSM. That work was reported to be done on their website in late 2015, however according to a report from the ALZ Forum, company president Craig Rosenfeld expects the trials to begin in April 2016. ALZ Forum also reported that the Phase 1 study will enroll between 30-50 people with ALS between trial sites in Melbourne and Syndey. People with both familial and sporadic forms of ALS will be enrolled and be part of a dose ranging study for approximately 1 month. We will add this study to our global clinical trial list when the full study design becomes available. Those interested are encouraged to register for our monthly clinical trial update by clicking here.
Helpful Links