Silence, please? Cells rely on enzymes called histone deacetylases (HDACs) to turn off the expression of certain genes. Video: University of Rhode Island.
Alterations in nearly 20 genes have been linked to ALS. But according to a growing number of studies, chemical changes to the genome might also be contributing to the disease.
Scientists are now looking to emerging cancer medicines in hopes to reset many of these so-called epigenetic switches in people with ALS. But the drugs tested to date appear to be ineffective in the clinic. Some researchers suspect that these medicines simply cannot be given at high enough doses to be effective. Their amounts strictly limited due to potentially harmful side effects.
Now, a growing number of scientists are developing so-called “next-generation” histone deacetylase (HDAC) inhibitors in hopes to create safe and more effective epigenetic medicines. More selective drugs are currently being tested in the cancer clinic. And, central nervous system (CNS)-available medicines are being developed for the treatment of neurodegenerative diseases.
This month, experts met up at Discovery on Target 2012 to share the latest epigenetic therapeutic strategies and their potential in the clinic going forward.
The meeting, which took place on October 1-2 2012 in Boston, highlighted emerging medicines for a wide-range of medical conditions including diabetes, sickle cell anemia and cardiovascular disease.
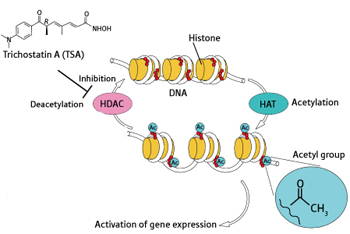
Overcoming obstacles Researchers first looked at general histone deacetylase inhibitors including trichostatin A in hopes to boost levels of neuroprotective substances in people with ALS. Image: Riken Institute, Japan.
Researchers first looked to epigenetic strategies in the late 1970s to treat cancer – hoping to stop the disease in its tracks by switching on tumor suppressive and tumor-killing genes. General HDAC-targeting medicines appeared promising in preclinical studies. But concerns of heart problems slowed their entrance into the clinic. Only two HDAC blockers are currently FDA-approved for use and only for the treatment of a rare form of the disease.
“The field is littered with failures,” says oncologist Kapil Dhingra MBBS. “Let’s call it a failure of blind enthusiasm.”
The field, however, appears to be turning a corner according to Dhingra, former head of Roche Oncology. A growing number of emerging HDAC-targeting medicines appear to be safe and exhibit “acceptable” toxicity profiles. But drug developers still remain in “phase II exploration mode” – looking largely for optimal combinations with other medicines to treat specific forms of the disease.
For researchers developing treatments for neurodegenerative conditions, however, the generation of safe and effective medicines are not the only challenges according to Repligen’s Senior Director of Preclinical Development Vincent Jacques PhD. These medicines need to be efficiently and effectively delivered into the central nervous system. “Most histone deacetylase inhibitors on the market today do not cross the blood brain barrier,” explains Jacques.
To meet these challenges, a growing number of scientists are looking to specific structural classes of histone deacetylase inhibitors such as benzamides which appear to be relatively safe, tolerable and can get into the brain and spinal cord.
Broad Institute's Stanley Center Director of Medicinal Chemistry Edward Holson PhD introduced drugs that appear to be primarily targeting HDAC3 that might be helpful in Alzheimer’s disease according to preliminary preclinical studies presented at DOT 12. And, Repligen’s Vincent Jacques PhD presented a clinical update on their lead candidate, RG2833, targeting HDAC1 and HDAC2 which currently is in phase I clinical trials for Friedrich’s ataxia. Whether a next-generation pan or selective HDAC-targeting medicine is the best choice is an open question and remains hotly debated in the field.
No study published to date has evaluated the benefits of any of these emerging medicines in ALS.
Tubastatin A Researchers are developing derivatives of HDAC6 blocker tubastatin A in hopes to generate medicines that help prevent transplant rejection and/or complications.
Protect and serve
Researchers first looked toward epigenetic medicines as a potential treatment for ALS in hopes to boost the production of potentially ALS-slowing neuroprotective substances. But a growing number of scientific studies suggest that inhibiting these histone-modifying enzymes might do much more to help fight the disease.
Treatment with HDAC6 blockers might “wake up” certain populations of regulatory T cells (Tregs), according to Children’s Hospital of Philadelphia (CHOP) organ transplant pathologist Wayne Hancock MBBS PhD, reducing inflammation. Elimination of HDAC6 appears to prevent transplant arteriosclerosis, a major life-threatening complication of heart transplants that occurs post-surgery according to preclinical studies Hancock presented at DOT12. What’s more, treatment with HDAC6 inhibitors appears to boost Treg function and protect mouse models against heart transplant rejection. Now, studies are underway at CHOP to determine if these same medicines can also help prevent heart transplant complications.
“These are the cells that keep you on the straight and narrow,” explains Hancock.
Including potentially people with ALS. Recent studies from Houston’s Methodist Hospital neurologist Stan Appel MD found that Tregs appear to help protect the motor nerves from destruction – reducing microglial-based neuroinflammation and extending survival in mouse models of disease. What’s more, the larger the numbers of circulating Tregs in people with ALS, the lower the progression rate of their disease according to a 2011 study. Treating people with ALS with HDAC6-targeting medicines therefore might boost the abilities of these cells to help keep neuroinflammation in check and thereby slow down the disease.
But according to Duke University School of Medicine research scientist Tso-Pang Yao PhD, the generation of HDAC4 inhibitors still remains a high priority – particularly for those developing treatment strategies for ALS. The deacetylating enzyme appears to be turned up in ALS and activates the production of proteins in skeletal muscle - resulting in muscle atrophy according to his studies. Potential HDAC4 blockers therefore might boost muscle strength in people with the disease.
To learn more about emerging medicines that may help reset chemical switches in people with ALS, check out our recent feature: Breaking the code of silence in ALS. To find out about the emerging role of T cells in ALS, tune into our podcast Symphony in M with Houston Methodist Hospital neurologist Stan Appel MD.
References
Beier, U.H., Wang, L., Han, R., Akimova, T., Liu, Y., Hancock, W.W. Histone deacetylases 6 and 9 and sirtuin-1 control Foxp3+ regulatory T cell function through shared and isoform-specific mechanisms. Science Signaling 5(229), ra45. Abstract | Full Text (Subscription Required)
Kalin, J.H., Butler, K.V., Akimova, T., Hancock, W.W. and Kozikowski, A.P. (2012) Second-generation histone deacetylase 6 inhibitors enhance the immunosuppressive effects of Foxp3+ T-regulatory cells. Journal of Medicinal Chemistry 55(2), 639-651. Abstract | Full Text
de Zoeten, E.F. et al. (2011) Histone deacetylase 6 and heat shock protein 90 control the functions of Foxp3(+) T-regulatory cells. Molecular and Cell Biology 31(10), 2066-2078. Abstract | Full Text
Beers, D.R., Henkel, J.S., Zhao, W., Wang, J., Huang, A., Wen, S., Liao, B. and Appel, S.H. (2011) Endogenous regulatory T lymphocytes ameliorate amyotrophic lateral sclerosis in mice and correlate with disease progression in patients with amyotrophic lateral sclerosis. Brain 134, 1293-1314. Abstract | Full Text
Zhao W, Beers DR, Liao B, Henkel JS, Appel SH. (2012) Regulatory T lymphocytes from ALS mice suppress microglia and effector T lymphocytes through different cytokine-mediated mechanisms. Neurobiology of Disease 48(3), 418-428. Abstract | Full Text (Subscription Required)
Choi, M.C., Cohen, T.J., Barrientos, T., Wang, B., Li, M., Simmons, B.J., Yang, J.S., Cox, G.A., Zhao, Y. and Yao, T.P. (2012) A direct HDAC4-MAP kinase crosstalk activates muscle atrophy program. Molecular Cell 47(1), 122-132. Abstract | Full Text (Subscription Required)
Cohen, T.J., Waddell, D.S., Barrientos, T., Lu, Z., Feng, G., Cox, G.A., Bodine, S.C. and Yao, T.P. The histone deacetylase HDAC4 connects neural activity to muscle transcriptional reprogramming. Journal of Biological Chemistry 282(46), 33752-33759. Abstract | Full Text
Further Reading
Kazantsev, A.G. and Thompson, L.M. (2008) Therapeutic application of histone deacetylase inhibitors for central nervous system disorders. Nature Reviews Drug Discovery 7(10): 854-868. Abstract | Full Text (Subscription Required)