More than 30 potential ALS medicines are being tested in the clinic today. Only Sanofi's Rilutek (riluzole), however, is FDA-approved to treat ALS. And, extends life 2-3 months.
Researchers are working hard to develop a more effective treatment for ALS. In 2013 alone, at least 10 new therapies entered the clinic.
Clinicians gathered at the 2013 annual meeting of the Northeast ALS (NEALS) Consortium to discuss the latest potential treatment strategies for ALS and the challenges implementing them going forward.
Neuralstem
Stemming the ALS tide? Neuralstem aims to stem the loss of motor neurons in people with ALS by boosting levels of neuroprotective substances. The potential therapy requires anti-rejection medicines. Video: Lane Niles PhD, Sanford-Burnham Medical Research Institute.
A phase II clinical trial of Neuralstem’s potential stem cell therapy for ALS is “now on” according to Emory University’s Jonathan Glass MD.
Sites include Emory University and University of Michigan. 15 people with ALS are expected to participate.
Neural stem cells, derived from a human embryo, will be surgically introduced into the cervical (diaphragm-moving) region of the spinal cord of people with ALS in hopes to protect motor neurons needed for breathing. At least 200,000 neural stem cells will be injected in 10 places throughout the C3 – C5 region on one or both sides of the spinal cord.
“We think the cells don’t migrate far,” explains Glass. “We think we can cover the whole pool of motor neurons with 10 injections.”
The study aims to determine the maximum number of stem cells that can be safely administered to potentially treat ALS.
Participants will also be checked on a regular basis to look for signs of reduced decline in functional abilities including breathing.
“We think the stem cells are getting in,” says Glass. “The question is what they are doing.”
To learn more about stem cell therapies for people with ALS and the challenges toward bringing them into the clinic, check out ISSCR 2013: ALS, Stem to Stern.
NeuRX DPS
A phase II clinical trial of Synapse Biomedical’s NeuRx diaphragm pacing system (DPS) is now underway at the State University of New York School of Medicine.
The device, developed by Case Western School of Medicine’s Ray Onders MD, aims to help people with ALS keep breathing longer by electrically conditioning the respiratory muscles.

DPS testing made easier? Neuromuscular ultrasound may soon enable clinicians to identify people with ALS eligible to receive the NeuRX DPS - without the discomfort and pain often associated with EMG and NCS.
The clinical trial aims to determine whether the NeuRx DPS improves diaphragm function of people with the ALS.
The study is to take place at 18 sites throughout the US. 180 people with ALS with breathing difficulties (FVC: 45-50%) are expected to participate.
A key goal of the clinical trial according to California Pacific Medical Center’s Jonathan Katz MD is to identify key respiratory signs (parameters) that might help clinicians identify people with ALS most likely to benefit from the procedure.
The pre-operative testing is extensive according to Duke University School of Medicine’s Rick Bedlack MD PhD. And, these tests do not necessarily reflect the ability of the respiratory muscles to be conditioned according to Cedar Sinai Medical Center’s Bob Baloh MD PhD. About one out of every five people with ALS wakes up in the recovery room without the device because their diaphragm cannot be stimulated according to preliminary results presented by his team at NEALS 2013.
What’s more, it remains unclear from these tests which people with ALS may benefit most from the device.
But this study is easier said than done according to Katz. The NeuRx DPS is approved by the FDA for humanitarian use. And, people receiving the standard of care (non-invasive ventilation) know that they are not being treated by the device.
“We knew that this study wasn’t going to be perfect,” says Katz.
To learn more about the NeuRX DPS, check out Clearing the air on the DPS?
***
Gilenya
A phase IIA clinical trial of Novartis’ Gilenya is now underway at Massachusetts General Hospital and Houston Methodist Hospital in Texas.
Regulating ALS? Immunomodulators including Gilenya aim to slow ALS by reducing inflammation. Image: National Institute of Allergy and Infectious Diseases.
The immunomodulator, currently used to treat multiple sclerosis, aims to slow progression of ALS by reducing infiltration of effector T cells, key instigators of inflammation – further damaging the motor nerves.
Gilenya may also increase circulation of regulatory T cells, key watchdogs that might help keep inflammation in check in people with ALS – at least early in the disease.
The study, led by Massachusetts General Hospital's James Berry MD MPH, aims to determine the safety and tolerability of Gilenya.
The one month clinical trial is to take place at 4 sites throughout the US. 30 people with ALS are expected to participate.
People with ALS will be monitored during the first day of the study for signs of a brief drop in heart rate. The potential treatable complication, known as bradycardia, according to Berry occurs in less than 0.5% of people first taking the medication.
“We need to be cautious about approaching this [strategy],” says Berry.
To learn more about Gilenya, check out our topics page.
Actemra
Elsewhere in the US, clinicians are gearing up to put Roche’s Actemra (tocilizumab)to the test in ALS. The immunomodulator, currently used to treat rheumatoid arthritis, aims to slow progression by reducing production of pro-inflammatory substances that might further damage the motor nerves.
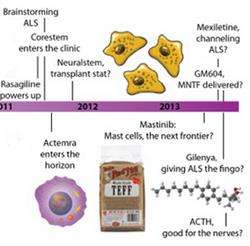
What's in the pipeline? Learn about therapies being developed for ALS in the clinic today by exploring our timeline.
The phase II clinical trial, announced at NEALS 2013 by MGH neurologist Merit Cudkowicz MD, is to be led by Barrow Neurological Institute’s Shafeeq Ladha MD and University of Kansas Medical Center’s Rick Barohn MD.
The study aims to determine the safety and tolerability of Actemra in people with ALS.
This is important according to Duke University School of Medicine's Tso-Pang Yao PhD because IL-6 signaling, blocked by Actemra, may also be needed to repair and regenerate damaged muscles in people with ALS.
Sites include the Barrow Neurological Institute in Arizona, University of California Los Angeles and the University of Kansas. 80 people with ALS are expected to participate.
To learn more about Actemra, check out our topics page.
CellCept, Methylprednisolone, Prograf, Prednisone and Simulect
Meanwhile, clinicians at Emory University School of Medicine are taking another look at immunosuppressants as a potential treatment for the disease.
The anti-rejection drugs, which include Genentech’s CellCept (mycophenolate mofetil) and Astella’s Prograf (tacrolimus), are the same medicines prescribed to people with ALS participating in the ongoing clinical trial of Neuralstem’s potential stem cell therapy for ALS.
The approach stems from one participant, known as “patient 11”, who appears to benefit from the transplantation procedure.
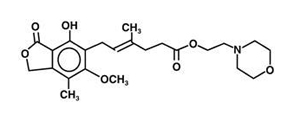
Suppress ALS? Some clinicians suspect that one person with ALS may appear to benefit from Neuralstem's stem cell-based treatment strategy because of anti-rejection medicines.
This benefit, however, according to Emory University School of Medicine’s Christina Fournier MD, may instead be due to the anti-rejection medicines. The reason, according to Fournier, is that this improvement appears to occur too rapidly to be explained by a potential stem cell treatment.
“We have to study this patient,” says Duke University School of Medicine’s Rick Bedlack MD PhD. “We have to figure out what in the world made him better."
A phase II clinical trial is to take place at Emory University School of Medicine, Massachusetts General Hospital and University of Massachusetts Medical Center. 30 people with ALS are expected to participate.
The multi-drug regimen includes intravenous injections of Novartis’ Simulect (basiliximab) and methylprednisolone during the first week. Decreasing doses of prednisone during the first month. And, Genentech’s CellCept and Astellas’ Prograf for 6 months.
“We might not understand how it works,” says Fournier. “So, we don’t want to change it.”
The study aims to identify another person with ALS that may also benefit from these medicines in hopes to determine why “patient 11” may benefit due to this treatment strategy.
These anti-rejection medicines, however, according to phase I results, are not tolerated by some people with ALS.
The phase II clinical trial is now ongoing. Initial results are expected sometime in 2015.
To learn more about potential immunotherapies for people with ALS including immunomodulators and immunosuppressants being tested for the disease, tune into our podcast with ALS TDI’s Steve Perrin PhD.
Exercise
Certain forms of exercise appear to be safe for people with ALS according to initial observations from an ongoing clinical trial led by Johns Hopkins University School of Medicine’s Nicholas Maragakis MD.

Exercise, does a body good? The Veterans Specific Activity Questionnaire (VASQ) may help clinicians create a safe exercise program for people with ALS according to results presented by Carolinas Medical Center physical therapist Mohammed Sanjak PhD PT MBA. Image: Northwest Guardian's Jim Bryant for Joint Base Lewis McCord, Washington.
The clinical trial is evaluating key forms of moderate aerobic and resistance exercise – stationary cycling and weightlifting - and comparing them to range of motion (ROM) exercises, the standard of care.
Sites include Carolinas Medical Center, Johns Hopkins University School of Medicine, Massachusetts General Hospital and Washington University School of Medicine.
The study aims to determine which forms of exercise are the most helpful for people with ALS.
All workouts are tailored to each person with ALS. No serious side effects were observed to date due to any of these exercise routines according to Johns Hopkins University School of Medicine’s Lora Clawson MSN CNRP.
To learn more about exercise and its potential benefits for people with ALS, check out Exercise: stretching the limits of ALS care.
Arimoclomol
Initial results from a phase II/III clinical trial of arimoclomol could be released as early as the fall of 2014 according to University of Miami’s Michael Benatar MBChB PhD.
Arimoclomol, developed by CytrX, aims to slow progression of ALS by reducing levels of misfolded superoxide dismutase I (SOD1). The buildup is a potential contributor of ALS – at least certain forms of familial disease.
***
To learn more about potential therapies being developed for ALS including elsewhere in the globe, check out our timeline ALS: In the pipeline 2013.
Patient Resources
A Dose Escalation and Safety Study of Human Spinal Cord Derived Neural Stem Cell Transplantation for the Treatment of ALS Contact | Clinical Trial | About Neuralstem
Note: Participants must live nearby either Emory University School of Medicine or University of Michigan. The reason according to Emory's Jonathan Glass MD is to ensure participants can be seen quickly as possible if complications develop due to anti-rejection medicines.
Diaphragm Pacing System (DPS) In Participants With ALS Contact | Clinical Trial | About the NeuRx DPS
Gilenya in ALS Contact | Clinical Trial | About Gilenya
Immunosuppression in ALS Contact | Clinical Trial
A Trial of Resistance and Endurance Exercise in ALS Contact | Clinical Trial
A Phase II/III Randomized, Placebo-controlled Trial of Arimoclomol in SOD1 Positive FALS Contact | Clinical Trial | About Arimoclomol
Note: This study is ongoing, but not recruiting.