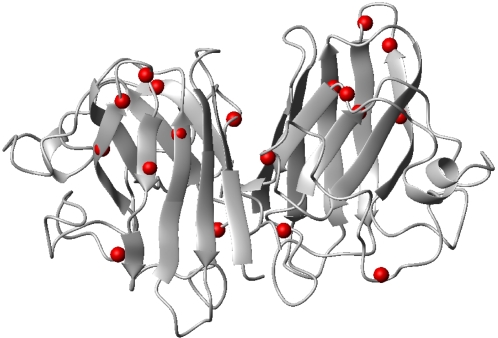
A free radical? As many as 1 out of 5 cases of familial ALS are caused by mutations (red) in the enzyme superoxide dismutase 1 (SOD1). Image: PLoS One.
Superoxide dismutase 1 (SOD1) is a key enzyme that helps keep our motor nerves healthy by detoxifying them. But in at least some people with ALS, these enzymes misfold- contributing to the disease.
Researchers remain unsure how misfolded SOD1 contributes to ALS. But according to studies led by University of British Columbia's Neil Cashman MD and University of Cambridge's Anne Bertolotti PhD, these misfolded enzymes may travel throughout the nervous system – driving the progression of the disease.
Scientists are working hard to develop potential medicines to reduce or eliminate misfolded SOD1 in people with ALS in hopes to slow or stop the disease in its tracks.
The antisense oligonucleotide SOD1Rx, developed by ISIS Pharmaceuticals, is currently at the phase I stage. Misfolded SOD1 antibodies, developed by Neurimmune Therapeutics AG and others licensed by Biogen Idec, are at the preclinical stage. Small molecules targeting misfolded SOD1, being developed by a team led by Neil Cashman MD and ALS Therapy Development Institute’s Fernando Vieira MD, are approaching the preclinical stage. And, misfolded SOD1 nanobodies, developed by University of Leuven's Wim Robberecht MD PhD, are beginning to emerge.
ALS Today’s Michelle Pflumm PhD talked with Neil Cashman MD, who developed the first antibody targeting misfolded SOD1, to learn more about this emerging strategy and its potential to treat ALS going forward.
Antibodies for sALS? Misfolded SOD1 antibodies may be of benefit to people with sporadic ALS according to a small study led by University of Massachusetts School of Medicine’s Daryl Bosco PhD. But the strategy remain hotly debated. Explore our timeline to learn about emerging strategies targeting SOD1 in ALS.
To find out more about SOD1 and ALS, explore our interactive timeline SOD1 at 20. To learn more disease progression in ALS, tune into our podcast with University of Pennsylvania School of Medicine’s Virginia Lee PhD and John Trojanowski MD PhD, Charting the course in ALS.
References
Grad, L. et al. (2014) Intercellular propagated misfolding of wild-type Cu/Zn superoxide dismutase occurs via exosome-dependent and -independent mechanisms. Proceedings of the National Academy of Sciences 111(9), 3620-3625. Abstract | Full Text
van Blitterswijk, M et al. (2011) Anti-superoxide dismutase antibodies are associated with survival in patients with sporadic amyotrophic lateral sclerosis. Amyotrophic Lateral Sclerosis 12(6), 430-438. Abstract | Full Text
Grad, L. et al. (2011) Intermolecular transmission of superoxide dismutase 1 misfolding in living cells. Proceedings of the National Academy of Sciences 108(39), 16398-16403. Abstract | Full Text
Munch, C., O’Brian, J., and Bertolotti, A. (2011) Prion-like propagation of mutant superoxide dismutase-1 misfolding in neuronal cells. Proceedings of the National Academy of Sciences 108(9), 3548-3553. Abstract | Full Text
Rakhit, R., Robertson, J., Vande Velde, C., Horne, P., Ruth, D.M., Griffin, J., Cleveland, D.W., Cashman, N.R. and Chakrabartty, A. (2007) An immunological epitope selective for pathological monomer-misfolded SOD1 in ALS. Nature Medicine 13(6), 754-759. Abstract | Full Text (Subscription Required)
Further Reading
Rotunno, M.S. and Bosco, D.A. (2013) An emerging role for misfolded wild-type SOD1 in sporadic ALS pathogenesis. Frontiers in Cellular Neuroscience 7, 253. Abstract | Full Text
Broering, T.J. et al. (2013) Identification of human monoclonal antibodies specific for human SOD1 recognizing distinct epitopes and forms of SOD1. PLoS One 8(4), e61210. Abstract | Full Text