Power gain. CK-357 may boost the power of fast skeletal muscles by increasing their sensitivity to weak electrical impulses generated by deteriorating motor nerves. CK-357 might be particularly useful to boost diaphragm function because these muscles contain nearly 50% fast-twitch muscle fibers. Courtesy of Nature Publishing Group. All Rights Reserved.
In people with ALS, the diaphragm and intercostal muscles gradually weaken often leading to respiratory distress and failure. In hopes to keep these muscles moving, Case Western University School of Medicine surgeon Raymond Onders MD FACS introduced a device, now approved by the FDA for people with ALS with breathing difficulties, called the NeuRX DPS which may boost the stamina of these muscles.
But researchers from San Francisco’s Cytokinetics Inc. think that they might have a simpler solution: the experimental drug CK-2017357 (CK-357). Introduced in 2008, CK-357 might increase the strength of certain skeletal muscles including those needed for breathing. Now, Cytokinetics scientists reveal just how CK-357 works: the drug promises to make the most of weakening neuromuscular junctions by helping fast twitch fibers in skeletal muscles hold on to calcium, enabling more powerful contractions. These so-called fast skeletal muscles are needed in part, to maintain a healthy breathing rate.
The Cytokinetics’ team anticipates that the drug could be beneficial in the treatment of number of neuromuscular diseases including ALS.
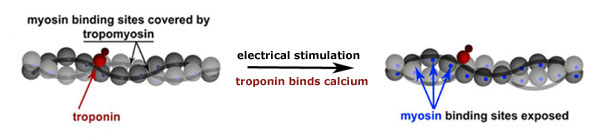
Sensitive muscle? Electrical signals from the motor nerves enable skeletal muscles to move by pulling the wrench out of the muscular works - troponin - through the release of calcium. CK-357 may strengthen muscles by increasing the affinity of fast skeletal troponin for calcium, boosting the power of these contractions. Adapted from Nature Education. Original source: Lehman, W. et al. (1994). Courtesy of Nature Publishing Group. All Rights Reserved.
Performance testing
Physicians are currently evaluating the safety and tolerability of multiple doses of CK-357 in people with ALS. The multi-institutional US team, led by State University of New York neurologist Jeremy Shefner MD PhD, are also checking for improvements in patients’ muscle function including breathing ability. The two 14 day placebo-controlled phase II clinical trials are expected to be completed by the end of March 2012. About 48 ALS patients are participating.
Meanwhile, researchers in England and France are gearing up to put the NeuRX DPS to the test to determine whether the device improves the quality of life and extends survival of people with ALS. The first results of these clinical trials are expected in early 2015.
Reference
Russell, A.J. et al. (2012) Activation of fast skeletal muscle troponin as a potential therapeutic approach for treating neuromuscular diseases. Nature Medicine doi:10.1038/nm.2618. Abstract | Full Text (Subscription Required)
Further Reading
Hardiman, O., van den Berg, L.H. and Kiernan, M.C. (2011) Clinical diagnosis and management of amyotrophic lateral sclerosis. Nature Reviews Neurology 7(11), 639-649. Abstract | Full Text (Subscription Required)
Hardiman, O. (2011) Management of respiratory symptoms in ALS. Journal of Neurology 258(3), 359-365. Abstract | Full Text (Subscription Required)
Patient Resources
Please note: These clinical trials are ongoing but are not recruiting.
A Study to Evaluate the Effects of Multiple Doses of CK-2017357 in Patients With Amyotrophic Lateral Sclerosis (ALS) ALSTDI | Website | Contact
Dose Titration Study to Test Safety and Effects of CK-2017357 in Patients With Amyotrophic Lateral Sclerosis (ALS) ALSTDI | Website | Contact