Breathe easier? Tirasemtiv may improve breathing in some people with ALS according to initial phase IIB results. But the drug did not appear to slow progression of the disease, the primary endpoint of the study. Courtesy of Nature Publishing Group.
Cytokinetics’ tirasemtiv may help some people with ALS breathe better according to initial phase IIB clinical trial results presented this week at the 2014 meeting of the American Academy of Neurology in Philadelphia.
The randomized placebo-controlled study found that people with ALS taking tirasemtiv for 12 weeks appeared to experience one third the reduction of breathing (slow vital) capacity (P < 0.0006) compared to those taking the placebo.
No significant improvement, however, was detected in other respiratory measures including an emerging indicator of strength of the diaphragm muscles (SNIP) – at least in people with lung disease indications.
The clinical trial, led by State University of New York neurologist Jeremy Shefner MD PhD, took place at 75 sites in 8 countries worldwide. 711 people with ALS participated. 473 people with ALS completed the study.
”This is the most positive ALS trial since riluzole,” says Shefner.
Cytokinetics introduced tirasemtiv in April 2008 in hopes to improve muscle function in people with neuromuscular diseases.
The drug, originally known as CK-2017357 (CK-357), aims to make the most of deteriorating neuromuscular junctions (muscle-motor nerve connections) by amplifying the signals generated by motor neurons that ‘tell’ the muscles to move.
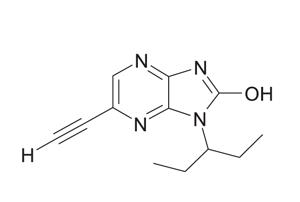
Sensitivity training? Tirasemtiv aims to keep muscles moving longer by increasing their sensitivity to weakening electrical impulses emitted by diseased motor nerves.
Tirasemtiv entered clinical testing in 2009. The potential muscle booster is currently being evaluated as a treatment of a growing number of neuromuscular diseases including myasthenia gravis.
The drug, according to phase IIB results, also appears to significantly improve the strength of certain skeletal muscles in the elbows, wrists, knees and ankles.
People with ALS taking tirasemtiv appeared to experience one third the reduction in muscle strength (P < 0.0158) vs those on placebo according to a so-called muscle megascore.
“Tirasemtiv may actually make patients stronger,” says participating investigator Bob Miller MD of California Pacific Medical Center (CPMC) in San Francisco. “Not just slow the decline.”
Tirasemtiv is the first drug to show a potential benefit at the phase II stage on respiratory and muscle function in people with ALS.
“We have for the first time evidence that a drug may improve breathing and muscle strength,” says participating investigator Merit Cudkowicz MD MSc of Massachusetts General Hospital (MGH).
A GI Bill?
Key challenges remain.
Tirasemtiv appears to be relatively safe. But according to phase IIB results, the drug is not well tolerated by many people with ALS.
More than 100 people with ALS dropped out in the first week of the study due to the inability to tolerate 250 mg of tirasemtiv – 50% of the study dose. And, nearly three times the number of participants dropped out of the clinical trial vs those on placebo.
Common side effects included nausea, dizziness, headaches and fatigue. People with ALS unable to tolerate tirasemtiv lost “slightly more” than twice the weight of those taking placebo.

Gathering strength Tirasemtiv may help keep certain skeletal muscles stronger longer according to phase IIB results. But the drug did not improve muscle fatigue - at least in the hands.
”I think further development of tirasemtiv depends on whether the tolerability of tirasemtiv can be improved,” says Shefner.
Mixed signals
In 2010, Cytokinetics began testing the potential muscle booster tirasemtiv in people with ALS in hopes to improve respiratory strength. Reduce fatigue. And, to help people with ALS do more everyday tasks.
But although some people taking tirasemtiv appeared to breathe better (as determined by breathing capacity) and certain muscles appeared stronger (as determined by muscle mega score), no significant improvement was detected in disease progression estimated by the ALS revised functional rating scale (ALSFRS-R), the primary endpoint of the phase IIB clinical trial.
“The ALSFRS-R is a great global measure of functional impairment in ALS.” says CPMC’s Bob Miller MD. “But, to me it is a very high bar. No drug as of yet has shown in a clinical trial an impact on that scale.”
Side effects including nausea, fatigue and loss of appetite may make it more difficult to assess the functional abilities of people with ALS taking tirasemtiv according to MGH’s Merit Cudkowicz MD.
“I think this may be more of a tolerability issue. The ALSFRS-R is very dependent on how patients feel,” says Cudkowicz.
And, a significant loss of weight may also be confounding efficacy analysis says SUNY neurologist Jeremy Shefner MD PhD.
The sniff test Sniff inspiratory pressure (SNIP) is an emerging measure that is being increasingly used to measure diaphragm strength in people living with chronic lung diseases. The measure according to CPMC's Bob Miller MD is relatively new to the ALS field and much less understood in terms of disease course than vital capacity.
More studies will be needed to determine whether tirasemtiv has the potential to slow progression of the disease.
People taking tirasemtiv did however appear to show potential improvements in strength of some skeletal muscles – at least determined by SVC and muscle megascore.
These so-called secondary outcomes, however, were also planned to be measured says Cytokinetics Chief Medical Officer Andrew Wolff MD. And, were part of the study protocol – the phase IIB clinical trial design.
“These are pre-specified secondary endpoints, they are not measures we looked at in a post-hoc manner,” says Wolff.
Promoting Tolerance?
Side effects of tirasemtiv, however, remain a considerable obstacle.
Only 50% of people with ALS taking tirasemtiv could tolerate 500 mg daily of tirasemtiv for 12 weeks according to principal investigator Jeremy Shefner MD PhD. 1 out of 4 participants taking tirasemtiv could tolerate only 250 mg daily – 50% of the study dose.
“We want to understand issues related to tolerability - how that can be improved,” says Cytokinetics’ CEO Robert Blum. “We need to delve more deeply into the [phase IIB clinical trial] data to know what we may do next.”
But MGH’s Merit Cudkowicz MD remains undaunted.
“We learned the side effect profile [of tirasemtiv] in the study,” says Cudkowicz. “They are all manageable.”
Medicines are available to reduce key gastrointenstinal (GI) side effects of tirasemtiv including nausea according to Cudkowicz. And, nutritional-based interventions could help people with ALS taking tirasemtiv maintain their weight.
The drug could also be introduced more slowly to people with ALS by ramping up the dose of tirasemtiv during a longer period of time says SUNY neurologist Jeremy Shefner MD PhD.
”I continue to feel that there is something here. And, so do my patients,” says CPMC’s Bob Miller MD.
***
To learn more about tirasemtiv and ALS, check out CK-357, helping PALS live strong? To find out more about the clinical trial including phase IIA results, read Tirasemtiv phase IIB powers up. To learn about other emerging strategies to help people with ALS breathe better, check out Clearing the air on the DPS?
Patient Resources
Study of Safety, Tolerability & Efficacy of CK-2017357 in Amyotrophic Lateral Sclerosis (ALS) (Completed) Contact | ALS TDI | Website
References
Hansen, R. et al. (2014) Tirasemtiv amplifies skeletal muscle response to nerve activation in humans. Muscle and Nerve, doi: 10.1002/mus.24239. Abstract | Full Text (Subscription Required)
Shefner, J. et al. (2013) A study to evaluate safety and tolerability of repeated doses of tirasemtiv in patients with amyotrophic lateral sclerosis. Amyotrophic Lateral Sclerosis and Frontotemporal Degeneration 14(7-8), 574-581. Abstract | Full Text (Subscription Required)
Shefner, J.M., Wolff, A.A. and Meng, L. (2013) The relationship between tirasemtiv serum concentration and functional outcomes in patients with ALS. Amyotrophic Lateral Sclerosis and Frontotemporal Degeneration 14(7-8), 582-585. Abstract | Full Text (Subscription Required)
Russell, A.J. et al. (2012) Activation of fast skeletal muscle troponin as a potential therapeutic approach for treating neuromuscular diseases. Nature Medicine 18(3), 452-455. Abstract | Full Text