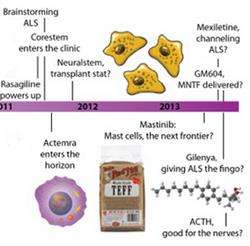
ALS Clinical Trials 101 Find out about therapies being developed for ALS in the clinic today by exploring our timeline.
More than 30 potential ALS medicines have been tested in the clinic. Only riluzole, however, is FDA-approved to treat ALS.And, increases survival 2-3 months.
Researchers are working hard to develop a more effective treatment for ALS. In 2012 and 2013 alone, more than 20 new strategies entered the clinic.
Ahead of the 2013 Northeast ALS (NEALS) Consortium meeting and the International Symposium on ALS/MND, ALS Today takes a second look at potential therapies for ALS being investigated in the clinic in this interactive timeline. Click on the pipeline to learn more about medicines being developed for the disease.
A long and winding road
In the 1980s, the first treatment of ALS emerged. Sanofi-Aventis’ riluzole, entering clinics in 1990, aims to help protect motor neurons by reducing glutamate buildup. The drug hit pharmacy shelves in 1995.
In 1993, scientists identified superoxide dismutase 1 (SOD1) as the first ALS-linked gene. And, began to develop treatment strategies to remove ‘free radicals’ in the brain and spinal cord. Mitsubishi Tanabe Pharma’s edavarone (Radicut), introduced in ALS clinics in 2001, aims to reduce the stress level of the motor neurons by lowering production of these toxic substances.
With the advent of the first mouse model of ALS, researchers discovered that the buildup of misfolded proteins may contribute to ALS. And, treatment strategies that aimed to sweep out aggregates out of motor nerves emerged. Chaperonin-targeted therapies including Cytrx’s arimoclomol aim to help proteins to properly fold. And, ISIS’ SOD1RX aims to reduce their synthesis by sopping up the RNAs via antisense-steeped sponges.
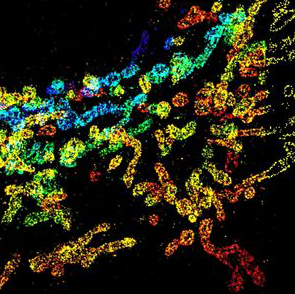
Power up? Mitochondrial-targeted medicines aim to slow progression of ALS by boosting power production in the motor nerves. Dexpramipexole and olesoxime however appeared to be ineffective in phase III trials. Image: Xiaowei Zhuang MD PhD, Harvard University.
By the late 1990s, ALS emerged as an energy crisis. And, clinicians began to look toward treatment strategies that could help keep the power on in the muscles and motor nerves. The metabolic supplement creatine, introduced in 2000, aims to boost energy production. And, putative mitochondrial-targeted therapies including Knopp’s dexpramipexole (licensed to Biogen Idec), Trophos’ olesoxime and Teva’s rasagiline (Azilect) hope to keep power production at full throttle by bolstering them.
But specific targets of ALS remained elusive. And, many research teams turned to more general strategies in hopes to develop treatments for the disease. Repurposed HDAC-targeted medicines aim to keep motor neurons healthy by throwing key genetic switches – turning up production of neuroprotective substances. And, stem cell strategies including Brainstorm’s NurOwn and Neuralstem’s NSI-266 hope to increase levels of these substances by delivering them directly to the nervous system.
In the mid 2000s, clinicians targeted the immune system in hopes to reduce neuroinflammation - slowing the progression of the disease. Anti-inflammatories including Sobi’s anakinra (Kineret) and Roche’s tocilizumab (Actemra) aim to soak up key substances that might damage the motor nerves in people with ALS. And more recently, immunomodulators including Novartis’ fingolimod (Gilenya) and Neuraltus’ NP001 may quiet down microglia by keeping out emerging instigators of inflammation including macrophages and certain T cells (Teffs).
Meanwhile, other research teams peered into the muscles of people with ALS in hopes to develop treatments for the disease. GlaxoSmithKline’s ozanezumab (anti-NogoA), entering clinics in 2009, aims to help keep the motor nerves connected. Cytokinetics’ tirasemtiv (CK-357) might help keep muscles moving by making the most of existing motor neuron-muscle connections. And, Synapse Biomedical's NeuRx diaphragm pacing system, introduced in 2007, hopes to help keep people breathing by conditioning the respiratory muscles.
Many of these potential medicines are being tested in the clinic today. Learn more about them by clicking on our interactive timeline.
Images: Courtesy of Children's Hospital of Philadelphia, Montreal Neurological Institute, National Institute of Neurological Disorders and Stroke, Paul Derry, Rockefeller University Press, Wikimedia Commons.
Patient Resources
A Clinical Trial of Nuedexta in Subjects with ALS Contact | ALS TDI | Website
A Study of Creatine Monohydrate in Patients With ALS Contact | ALS TDI | Website
A Phase 3 Study of MCI-186 for Treatment of ALS ALS TDI | Website
A Phase II/III Randomized, Placebo-controlled Trial of Arimoclomol in SOD1 Positive FALS Contact | ALS TDI | Website
A Study of Ozanezumab (GSK1223249) Versus Placebo in the Treatment of ALS ALS TDI | Website
A Study of Safety, Tolerability & Efficacy of CK-2017357 in ALS ALS TDI | Website
A Study of Rasagiline in Patients With ALS Contact | ALS TDI | Website
SOD1 Inhibition by Pyrimethamine in Familial ALS Contact | ALS TDI | Website
A Trial of Safety and Efficacy of Rasagiline in Patients With ALS Contact | ALS TDI | Website
Safety and Tolerability of Anakinra in Combination With Riluzol in ALS Contact | ALS TDI | Website
Autologous Cultured Mesenchymal Bone Marrow Stromal Cells Secreting Neurotrophic Factors (MSC-NTF), in Patients With ALS Contact | ALS TDI | Website
A Safety Study of HLA-haplo Matched Allogenic Bone Marrow Derived Stem Cell Treatment in ALS ALS TDI | Website
Dose Escalation and Safety Study of Human Spinal Cord Derived Neural Stem Cell Transplantation for the Treatment of Amyotrophic Lateral Sclerosis Contact | ALS TDI | Website
An Evaluation of masitinib in ALS Contact | ALS TDI | Website
Mexiletine in Sporadic ALS Contact | ALS TDI | Website
Mexiletine for the Treatment of Muscle Cramps in ALS Contact | ALS TDI | Website
Gilenya in Amyotrophic Lateral Sclerosis Contact | ALS TDI | Website
A Study to Explore the Safety and Tolerability of Acthar in Patients With ALS Contact | ALS TDI | Website
A GM604 Phase 2A Randomized Double-blind Placebo Controlled Pilot Trial in ALS Contact | ALS TDI | Website