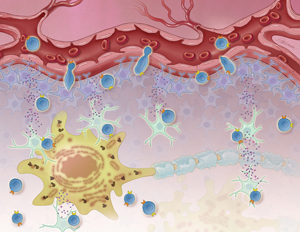
T mobile Certain cytotoxic T cells infiltrate the brain and spinal cord in people with ALS - potentially contributing to disease progression. Image: Jennifer Fairman for ALS TDI.
T cells help keep our bodies free from infection. But in people with ALS, some of these white blood cells appear to enter attack mode. And, unleash a storm of toxic substances that damage the motor nerves - fueling the progression of the disease.
Researchers hope to develop medicines that protect the motor nerves in people with ALS by keeping these troublesome T cells out of the nervous system. One of these emerging treatment strategies, fingolimod, locks them up in the lymph nodes.
The FDA-approved multiple sclerosis medicine, marketed by Novartis under the name Gilenya, is one of a growing number of immune system-based treatment strategies that hope to slow ALS by reducing inflammation. Others include Neuraltus Pharmaceuticals' NP001, Roche’s Actemra (tocilizumab) and UCB’s CDP7657 (anti-CD40L Fab).
The phase IIA clinical trial is expected to begin in the spring of 2013.
ALS Today’s Michelle Pflumm PhD talked to ALS TDI CEO and Chief Scientific Officer Steve Perrin PhD to learn more about Gilenya, other emerging immunomodulators for ALS and their potential to treat the disease going forward.
To learn more about emerging role of the immune system in ALS and potential treatment strategies for the disease, check out NP001, a quiet riot for ALS?