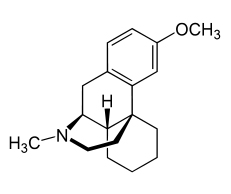
A dynamic duo? Nuedexta contains dextromethorphan (DM) (above) and a small amount of quinidine to slow DM breakdown.
One of the biggest fears for people with ALS is losing the ability to be speak. For many, this is a challenge that might be faced late into the disease. But for people with the bulbar-onset form of ALS, trouble speaking is amongst the first signs of the disease.
Now, US researchers are gearing up to evaluate a medicine that may help people with ALS retain some of this so-called bulbar control. The drug, called Nuedexta, is currently used in the US to help people, including those with ALS, to control their emotions.
Nuedexta is one of at least three medicines currently or soon to be evaluated that may improve the quality of life for people with ALS.
“If you can improve the quality of life,” says Center for Neurologic Study (CNS) neurologist Richard Alan Smith MD, leader of the study, “This represents real progress in tackling the disease and providing support for patients.”
The clinical trial is one of a number of studies within the growing “TREAT ALS” portfolio, an ALS Association initiative that helps push emerging ALS medicines into the clinic.
To speak, perchance to dream. Scientists suspect that DM may stimulate receptors that decorate motor neurons in the brainstem to help people speak and swallow. Adapted from Longone, P. et al. (2011).
CNS scientists first suspected in the 1990s that dextromethorphan (DM), a neuroprotective substance, might be helpful to people with ALS by slowing down the progression of the disease. But when people with ALS starting take the drug in a small phase I safety study, the team noticed something else. DM helped people with ALS control their emotions.
Subsequent phase II and phase III clinical trials in the early and mid 2000s found that the number of uncontrollable crying and laughing outbursts dropped about 50%.
The condition, known as pseudobulbar affect (PBA), occurs in about 20% - 50% of people with ALS. PBA is suspected to occur due to structural damage sustained by certain parts of the brain which control emotions.
The medicine, reformulated and named Nuedexta, was FDA-approved for use to treat PBA in October 2010.
Speak Up
Nuedexta, however, may help people with ALS do more than keep their emotions in check.
A growing number of people with ALS taking Nuedexta for PBA according to anecdotal observations also appear to show signs of improvements in the rate and quality of their speech and their abilities to swallow.
“Nuedexta can help reduce PBA. We know that,” says Duke University School of Medicine neuorologist Rick Bedlack MD PhD MS. "But what’s shocking is that the drug can, at least temporarily, improve [bulbar] dysfunction when upper motor neurons go bad.”
The drug, acting on receptors decorating motor neurons in the brainstem, may also be helping people with ALS maintain bulbar control.
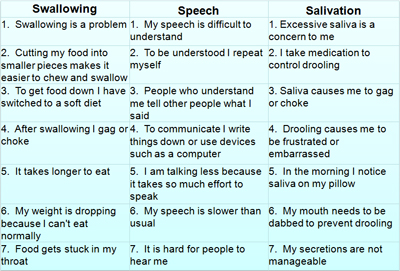
Measuring stick The CNS-bulbar function scale, a self-reported measure (above) co-developed by Patientslikeme, will be primarily used to estimate Nuedexta's ability to help people with ALS speak and swallow. Courtesy of Paul Wicks PhD. Reproduced with permission.
Now, the US team is gearing up to put Nuedexta to the test in people with ALS that are having trouble speaking and swallowing. The placebo-controlled randomized crossover clinical trial is to be conducted at 7 Northeast ALS Consortium (NEALS) sites in the US including Massachusetts General Hospital. All participants are to be treated with Nuedexta and placebo successively for 30 days separated by a 10-15 day washout period. Primary outcomes include: self-reported and clinician-assessed improvements in speech quality and swallowing abilities. Other measures include: ALS-FRS. 60 people with ALS are expected to participate.
The trial is scheduled to begin in the spring of 2013.
This is the first medicine to be tested specifically to help people with ALS speak and swallow.
“We can compensate for [abilities] people are losing but we have been unable to take these problems and make them better,” says Bedlack. “Now, for the first time, we are starting to see things like that.”
Patient Resources
Clinical Trial of Nuedexta in Subjects with ALS Contact | ALS TDI | Website
References
Pioro, E.P., et al. (2010) Dextromethorphan plus ultra low-dose quinidine reduces pseudobulbar affect. Annals of Neurology 68(5), 693-702. Abstract | Full Text
Brooks, B.R., et al. (2004) Treatment of pseudobulbar affect in ALS with dextromethorphan/quinidine: a randomized trial. Neurology, 63(8), 1364-1370. Abstract | Full Text (Subscription Required)
Al-Saif, A., Al-Mohanna, F. and Bohlega S. (2011) A mutation in sigma-1 receptor causes juvenile amyotrophic lateral sclerosis. Annals of Neurology, 70(6), 913-919. Abstract | Full Text (Subscription Required)
Mavlyutov, T.A., Epstein, M.L., Andersen, K.A., Ziskind-Conhaim, L. and Ruoho, A.E. (2010) The sigma-1 receptor is enriched in postsynaptic sites of C-terminals in mouse motoneurons. An anatomical and behavioral study. Neuroscience 167(2), 247-255. Abstract | Full Text
Further Reading
Rosen, H. (2008) Dextromethorphan/quinidine sulfate for pseudobulbar affect. Drugs of Today 44(9), 661-668. Abstract | Full Text
Update (3/10/13) : Clinical trial launch date rescheduled. Patient resources added.
Update (9/10/12) : Researchers recently revised the CNS-BFS scale. The figure has been updated accordingly.